A battery is a device which stores chemical energy and which can convert it into electricity. There are two types of batteries: primary batteries (single use) and secondary batteries (rechargeable), also known as accumulators.
In this post, I will explain how to chose the right batteries for your projects and how to compute how long will that battery last.
Primary Batteries (single use)
The primary batteries are based on irreversible electrochemical reactions, which makes them non-rechargeable. The primary advantage of these batteries is that they have a low self-discharge current and they’re used in applications which require long term energy storage.
The alkaline batteries are part of this category, they have a nominal cell voltage of 1.5 V and they offer an energy density of 85 Wh / kg, respectively 250 Wh / L (when related to the volume). Their self discharge rate is less that 0.3% / month.
Secondary Batteries (rechargeable)
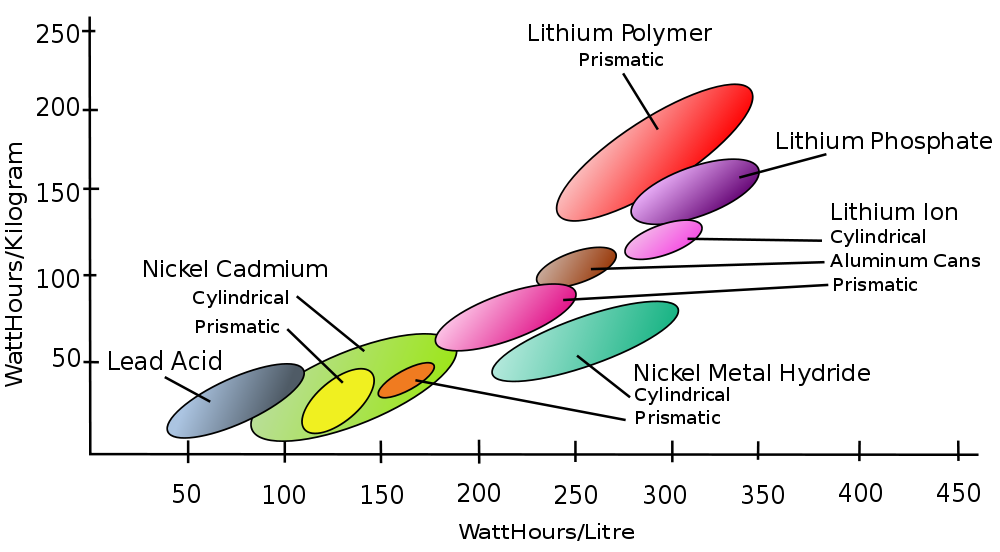
From this category, there are a few type of batteries worth mentioning: lead acid batteries, nickel metal hybrid (NiMh) batteries, lithium ion batteries (Li-Ion) and lithium ion polymer batteries (LiPo).
The lead acid batteries have a nominal cell voltage of 2.1 V and a quite low energy density (30-40 Wh/kg, respectively 60-75 Wh/L). They have a durability of 500-800 charge-discharge cycles and a time durability of 5-8 years (in automotive operating conditions).
The NiMh batteries have a nominal cell voltage of 1.2 V, an energy density of 30-80 Wh/kg, respectively 140-300 Wh/L, and a durability of 500-1000 cycles. The NiMh batteries have a high self-discharge current (about 30% / month).
The Li-Ion batteries have a nominal cell voltage of 3.6 V, an energy density of 150-250 Wh/kg (or 250-360 Wh/L), a durability of 1200-10000 cycles and a self discharge rate of 5-10% / month. The LiPo batteries have similar characteristics and a durability of 500-1000 cycles.
How much energy does the battery actually store ?
The batteries are usually rated in mAh (for example 1000 mAh). This rating actually measures the charge stored by the battery and it means that a 1000 mAh battery for example is capable to supply a current of 1000 mA for 1 h.
Knowing the nominal voltage and the rating of the battery, we can compute the amount of energy stored as follows:
\(E = V \cdot Q\)
Where:
\(E\) is the energy that the battery can store (measured in joules; \(1 J = 1 Ws = 1 VAs\))
\(V\) is the nominal voltage of the battery
\(Q\) is the charge that can be stored by the battery (measured in coulombs; \(1 C = 1 As\))
For example, a battery with a rating of 1000 mAh can store the following charge:
\(Q = 1000 \cdot 10^{-3} \cdot 3600 As = 3600 C\)
If this battery has for example nominal voltage of 3.7 V, then it stores the following energy:
\(E = V \cdot Q = 3.7 \cdot 3600 J = 13320 J\)
How long will the battery last ?
The amount of time that a battery lasts from a charge depends on the energy storage capacity of the battery and on the power consumption of the device that is powers.
You can compute the amount of time that a battery will last from a single charge as follows:
\(\large{t = \frac{E}{P}}\)
Where:
\(t\) is the amount of time that the battery will last from a single charge (measured in seconds)
\(E\) is the amount of energy that the battery can store (measured in joules)
\(P\) is the power consumption of device which will be powered from the battery (measured in watts; \(1 W = 1 VA\))
What type of battery to choose ?
Overall, the LiPo batteries are usually the best option, considering the energy density, the price and the durability. They come in a many form factors, from a single LiPo battery cell to multi-cell packs. The LiPo chargers are readily available, and for one cell and two cell LiPo batteries, there are some chargers available even as small electronic modules.